
Tokyo University of Science
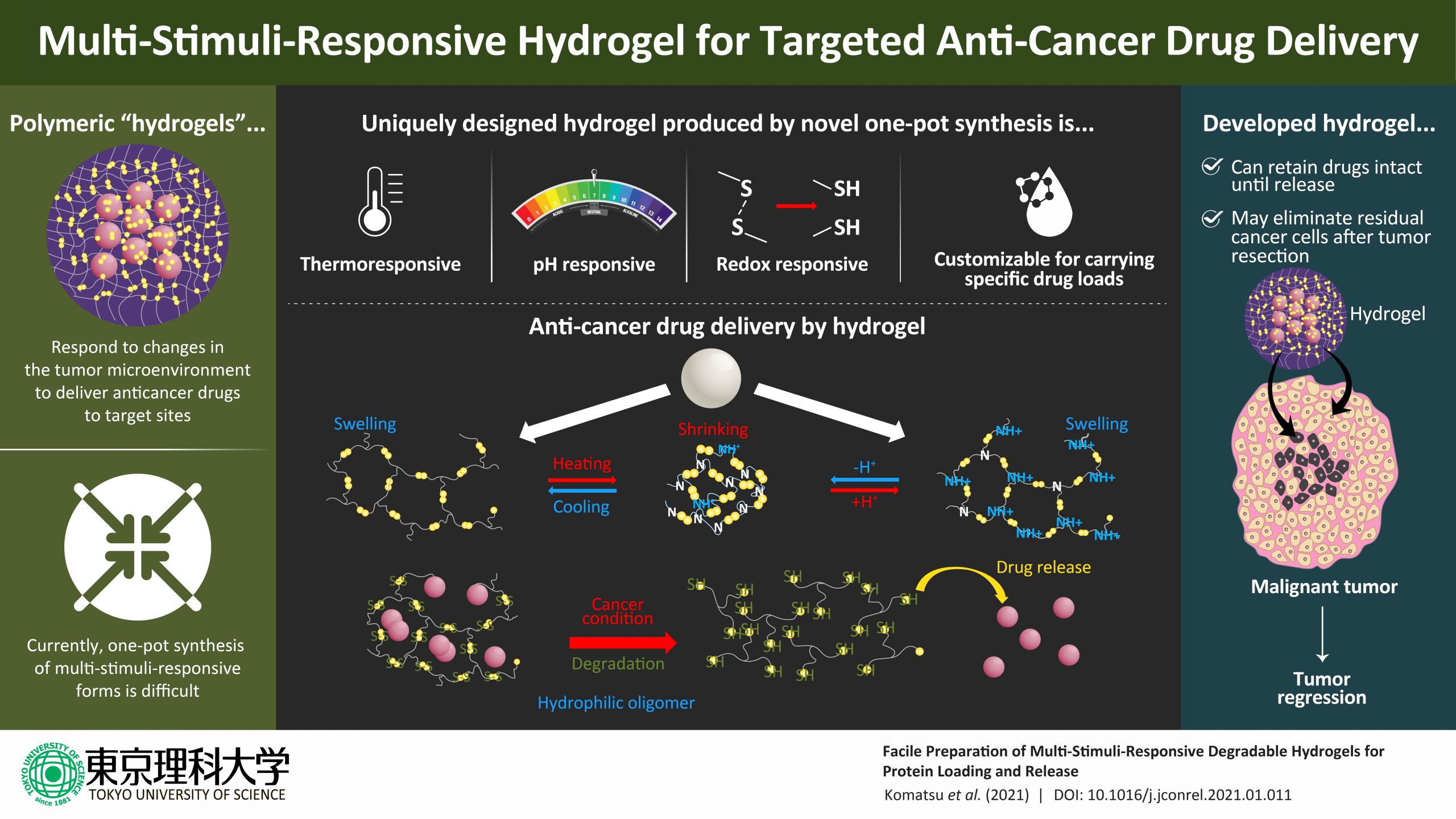
Hydrogels are often used as drug delivery systems, but to be effective carriers for anti-cancer drugs, they need to be responsive to varied stimuli in the tumour microenvironment. Now, scientists from Japan have developed novel hydrogels to effectively deliver drugs to tumour sites in response to temperature and pH changes in the tumour microenvironment.
These multi-stimuli-responsive hydrogels can eliminate remnant cancer cells following tumour excision through controlled drug release, offering hope for effective cancer treatment.
A team of scientists, led by Professor Akihiko Kikuchi from Tokyo University of Science, reports the production of unique degradable hydrogels that respond to changes under multiple conditions in “reducing” environments mimicking the microenvironment of tumors.
As Prof. Kikuchi observes, “In order to prepare degradable hydrogels that can release drugs in response to changes in the tumor microenvironment, we prepared hydrogels that respond to temperature, pH, and reducing environment, and analyzed their properties.”
In their study published in the Journal of Controlled Release, Prof. Kikuchi—along with his colleagues from Tokyo University of Science, Dr. Syuuhei Komatsu, Ms. Moeno Tago, and Ms. Yu Ando, and his collaborator on the study, Prof. Taka-Aki Asoh from Osaka University—details the steps of designing these novel hydrogels from the synthetic polymer poly(ethylene glycol) diglycidyl ether and the sulfur-containing organic compound cystamine. In response to low temperatures, these hydrogels swell up while they shrink at the physiological temperature.
Additionally, the hydrogels respond to pH changes by virtue of possessing tertiary amino groups. It must be noted here that the pH of the tumour microenvironment fluctuates between 5.5 and 6.5 owing to glycolysis in the tumour cells. Under the reducing conditions of this environment, the hydrogels degrade because of the breakage of disulfide bonds and change into low molecular-weight water-soluble oligomers that are easily excreted from the body.
To further test their drug release properties, the scientists loaded these hydrogels with specific proteins by exploiting their temperature-dependent swelling-deswelling behavior and tested the controlled release of drugs under acidic or reducing conditions.
It was found that the amount of drug loaded onto these hydrogels could be controlled by changing the mesh size of the hydrogel polymer network by changing temperature, suggesting the possibility of customizing these DDSs for specific drug delivery. Besides, the hydrogel network structure and electrostatic interactions in the network ensured that the proteins were preserved intact until delivery, unaffected by the swelling and shrinking of the hydrogels with pH changes in the surrounding environment.
The scientists found that the loaded protein drugs were completely released only under reducing conditions.
Using these hydrogels and the traceability that they provide, doctors may soon be able to design “customized” hydrogels that are specific to patients, giving personalized medicine a big boost. In addition to that, this new DDS provides a way to kill cancer cells that are left behind after surgery.
As Prof. Kikuchi states, “The implantation of this material in the affected area after cancer resection may eliminate residual cancer cells, making it a more powerful therapeutic tool”.
As cancer tightens its vice grip around the world, treatment options need to be varied and upgraded for customized and effective therapy. This unique and simple design technique to produce multi-stimuli-responsive hydrogels for effective drug delivery to target tumour sites may just be one among several such promising techniques to mount an answer to the challenge cancer poses to humanity.