
Muhammad Sheraz
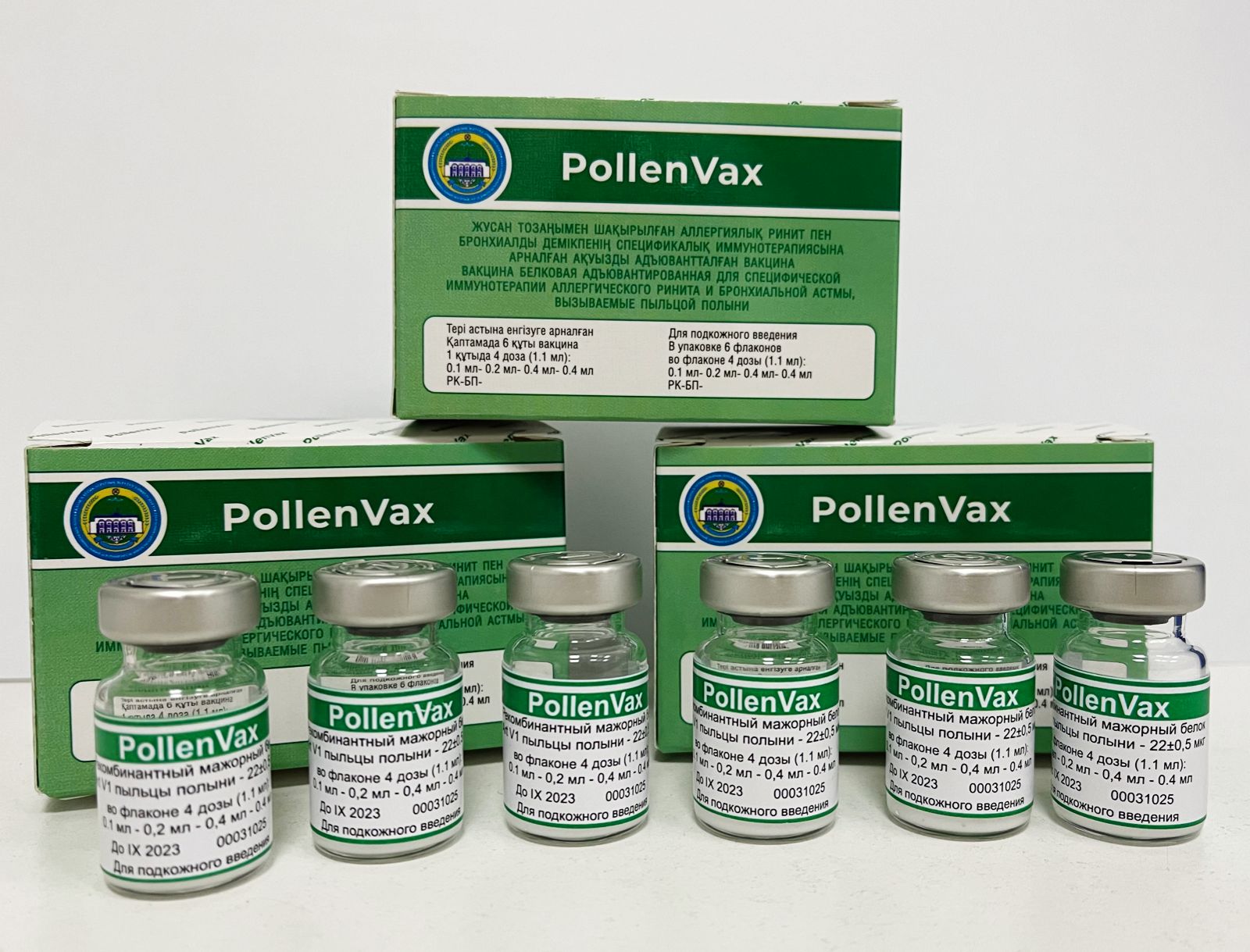
The International Center for Vaccinology (ICV) at Kazakh National Agrarian Research University (KazNARU) has developed a novel vaccine of immunotherapy for allergic rhinitis and bronchial asthma caused by wormwood pollen (one of the ten global aeroallergens).
ICV Director Professor Kaissar Tabynov is an expert in the field of vaccinology. His main specialization is associated with the development, study, and implementation of the production of various vaccines for animals, birds, and humans. He told us that the new Vaccine is based on a recombinant wormwood pollen major protein Art v 1 formulated with the oil adjuvant ISA-51 and it is named “PollenVax”.
The uniqueness of this new vaccine is that in an ultrashort ASIT regimen with four subcutaneous desensitizing immunizations (total course duration of one month) in a mouse model it provides pronounced therapeutic effectiveness against allergic rhinitis and asthma caused by wormwood pollen. (https://pubmed.ncbi.nlm.nih.gov/35371056/).“PollenVax vaccine potentially solves not only these disadvantages of ASIT, but also others related to the ASIT schedule, which, depending on the drug and type of allergy, can be continuous year-round, pre-season (before the allergy season begins) or combined pre/co-season (during the allergy season),” explained Professor Tabynov. Since in many countries, including Kazakhstan, the wormwood blooming period is long enough (from July to October), the possibility of conducting co-seasonal ASIT is of great practical importance, he said.
“PollenVax” vaccine in ultrashort pre- and co-seasonal ASIT when used in high and low-dose immunization regimens provides a significant reduction in both signs of allergic rhinitis and bronchial asthma in sensitized mice (https://pubmed.ncbi.nlm.nih.gov/36439113/). This vaccine is currently at NASA Technology Readiness Level 5 (TRL-5) according to the BIRAC scale for the biotechnology area (vaccines), as it has successfully passed pre-clinical safety studies (acute and chronic toxicity, allergenicity) in laboratory animals (mice, rats, guinea pigs) at the National Center for Drug and Medical Device Expertise of the Ministry of Health of the Republic of Kazakhstan (Regulatory Authority; Report No. TDI-2022/07 dated October 28, 2022).
In the laboratory, “PollenVax” vaccine demonstrated significantly superior therapeutic efficacy compared to the commercial drug CLUSTOID based on treated Artemisia vulgaris extract formulated with aluminum hydroxide adjuvant (ROXALL Medizin GmbH, Germany). “PollenVax” vaccine is recommended by the regulatory agency of Kazakhstan for further clinical trials.
“PollenVax” is not the first vaccine that KazNARU’s International Center for Vaccinology (ICV) has developed. Prior to this vaccine, the Center developed two Covid-19 vaccines “NARUVAX-C19” (subunit for intramuscular injection) and NARUVAX-C19/Nano (nano-vaccine for nasal spraying). NARU in vaccines names stands for ‘National Agrarian Research University’, VAX for vaccine and C19 for COVID-19 coronavirus, and Nano for nano-vaccine. Both vaccines successfully passed a series of safety, immunogenicity, and protective efficacy studies on different laboratory animals (mice and hamsters) at Almaty’s “Aikimbayev National Scientific Center for Especially Dangerous Infections” (NSCEDI). The NSCEDI has a Biosafety Level 3 laboratory for working with viruses (BSL-3). It is the only Center in Kazakhstan and Central Asia that has a Biosafety Level 3 laboratory for working with animals (ABSL-3).
Also, ICV Director Professor Kaissar Tabynov contributed to the development of three other vaccines for public health: 1: Refluvac - against pandemic influenza A / H1N1pdm09; 2: Kazfluvac - against pandemic influenza A / H5N1; 3: Kazfluvir - against seasonal influenza). Kazfluvir, which has successfully passed the III-phase clinical trial in Kazakhstan. Professor Tabynov and his team also developed seven vaccines for veterinary medicine (against highly pathogenic avian influenza H5 subtypes and H7, equine influenza, bovine, and small bovine brucellosis).