
Tokyo University of Science
The cells in our body are constantly fighting off the threat of cancer by repairing damaged DNA. In a new study, scientists from the Tokyo University of Science investigate the structure of an elusive protein complex that plays a key role in the activation of the “Fanconi anemia pathway” involved in DNA repair, and report on the factors governing its stability. Their insights can potentially help find novel treatments for disorders involving chromosomal instability, including cancer.
Associate Professor Tatsuya Nishino and his colleague Dr. Sho Ito from Tokyo University of Science decided to explore the crystalline structure of this intriguing complex using X-ray diffraction techniques.
“DNA damage and chromosome segregation are mechanisms necessary for the maintenance and inheritance of genes possessed by all organisms. MHF (also known as CENP-SX) is an enigmatic complex that plays a role in DNA repair and chromosome segregation. We wanted to find out how it performs these two different functions in the hope that it might give us insights into novel phenomena,” explains Prof. Nishino. Their findings are published in Acta Crystallographica Section F: Structural Biology Communications.
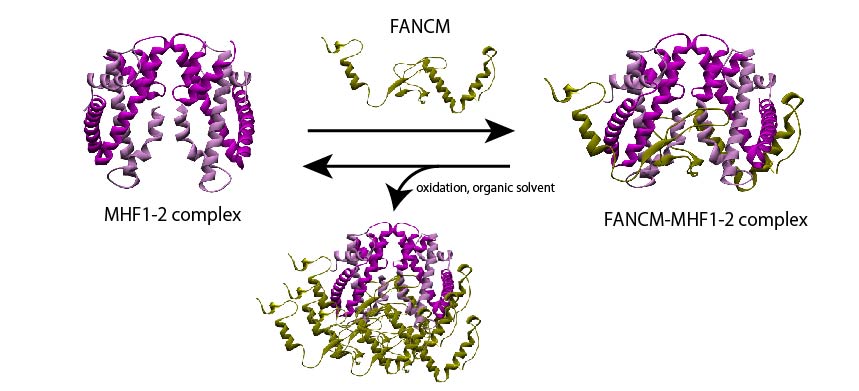
The scientists prepared a recombinant version of the FANCM-MHF complex, consisting of FANCM from chickens and MHF1 and MHF2. They were able to purify three different types of protein crystals—tetrahedral, needle-shaped, and rod-shaped—from similar crystallization conditions. Surprisingly, upon determining the structure with X-ray crystallography, they found that two of the crystal forms (tetrahedral and needle-shaped) contained only the MHF complex without FANCM.
Intrigued by this discovery, the scientists used biochemical techniques to examine what caused the FANCM-MHF complex to disassemble. They attributed it to the presence of a compound called 2-methyl-2, 4-pentanediol (MPD), an organic solvent commonly used in crystallography, and exposure to an oxidizing environment.
But, how exactly does the dissociation happen? The scientists believe that this may have been caused partly by certain non-conserved amino acids in the chicken FANCM which causes the complex to aggregate with other FANCM-MHF complexes and disassemble. Additionally, they surmise that the small, flexible structure of MPD may have also allowed it to bind to and facilitate the release of FANCM, dismantling the complex.
The findings are extraordinary and can be used to improve the stability of the FANCM-MHF complex for future studies on its structure and function. Dr. Ito believes we have much to expect in the future from this complex.
“A good understanding of this complex can help us treat cancer and genetic diseases, create artificial chromosomes, and even develop new biotechnological tools,” he speculates.