NCUSEC
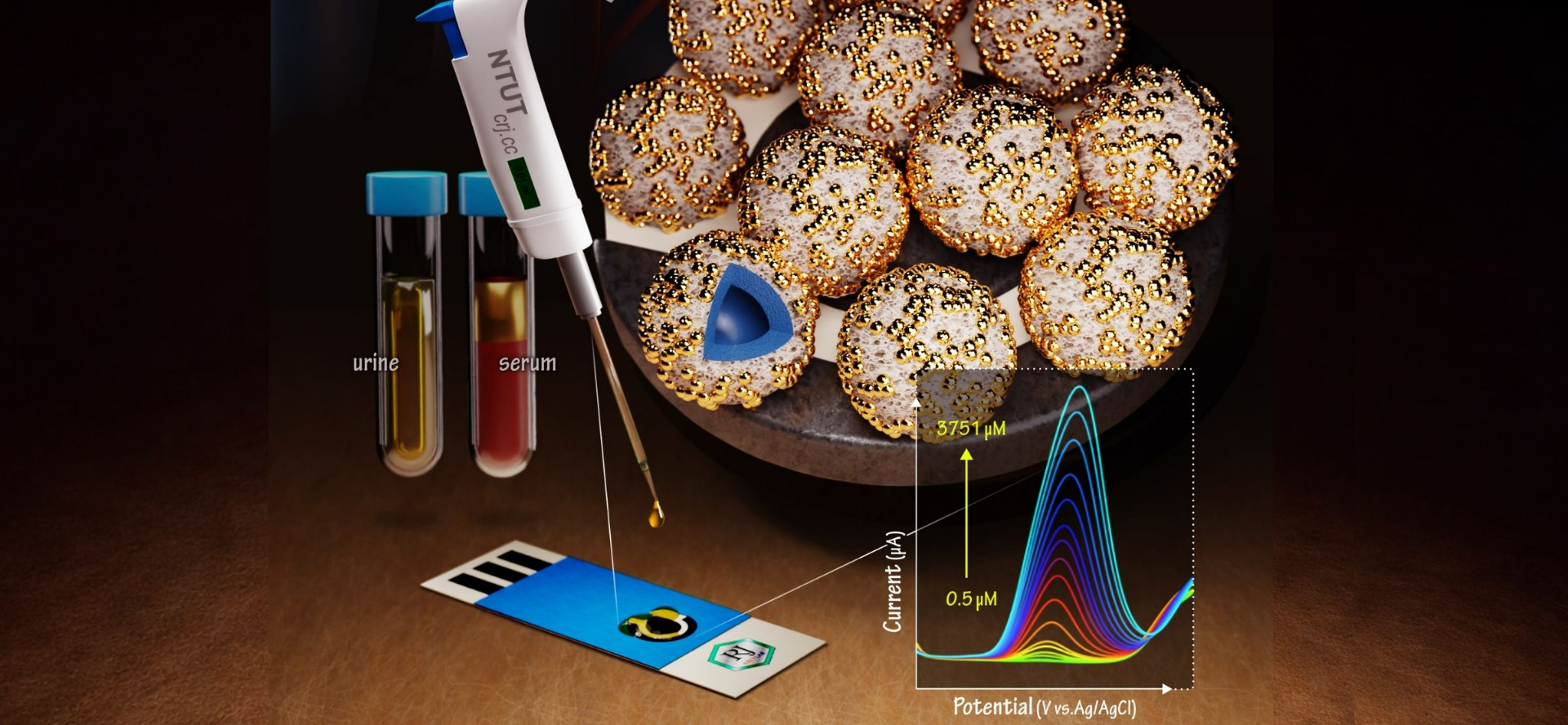
A collaborative research team led by Associate Professor Cihun-Siyong Gong from the Department of Electrical Engineering at National Central University (NCU) and Professor Ren-Jei Chung from the Department of Chemical Engineering and Biotechnology at National Taipei University of Technology has developed a green electrochemical sensing platform driven by caffeic acid. The study has been published in the internationally renowned journal ACS Sensors.
The researchers first synthesized self-templated, double-shelled zinc manganite (ZnMn₂O₄) hollow microspheres using co-precipitation and calcination methods. At room temperature, a natural organic molecule—caffeic acid—was employed as a reducing agent to enable the in situ formation and uniform deposition of rhenium nanoparticles on the shell surface, resulting in a Re@ZnMn₂O₄ composite material. This approach successfully avoids the use of highly toxic reducing agents, aligning with green chemistry principles, while the double-shelled structure provides a large accessible surface area and abundant active sites for reactions.
Material characterization revealed that the incorporation of rhenium nanoparticles enhances conductive pathways, significantly improving electron transport and interfacial reaction efficiency. Electrochemical testing further demonstrated that the electrode exhibits optimal performance under near-physiological conditions (pH 7).
In terms of sensing performance, the platform achieves a low detection limit of 0.21 μM for epinephrine, along with a wide linear detection range and high selectivity. Notably, the sensor retains approximately 98% of its signal response after 12 days of storage at room temperature, indicating excellent long-term stability.
Overall, this study integrates advanced structural design with green synthesis strategies to propose a high-performance electrochemical sensing approach. The findings lay a solid foundation for future real-time biomedical monitoring systems and highlight strong potential for clinical applications.